Heat And Thermodynamics Ques 139
- An ideal gas expands isothermally from a volume $V _1$ to $V _2$ and then compressed to original volume $V _1$ adiabatically. Initial pressure is $p _1$ and final pressure is $p _3$. The total work done is $W$. Then,
(2004, 2M)
(a) $p _3>p _1, W>0$
(b) $p _3<p _1, W<0$
(c) $p _3>p _1, W<0$
(d) $p _3=p _1, W=0$
Show Answer
Answer:
Correct Answer: 139.(c)
Solution:
Formula:
- Slope of adiabatic process at a given state $(p, V, T)$ is more than the slope of isothermal process. The corresponding $p-V$ graph for the two processes is as shown in figure.
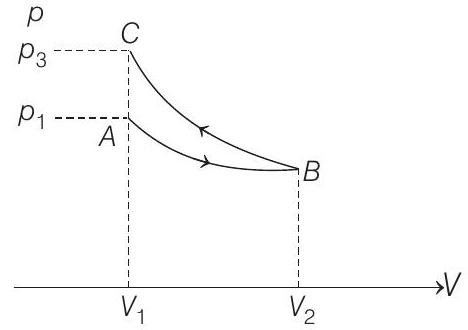
In the graph, $A B$ is isothermal and $B C$ is adiabatic.
$$ W _{A B}=\text { positive (as volume is increasing) } $$
and $W _{B C}=$ negative (as volume is decreasing) plus,
$\left|W _{B C}\right|>\left|W _{A B}\right|$, as area under $p$ - $V$ graph gives the work done.
Hence, $\quad W _{A B}+W _{B C}=W<0$
From the graph itself, it is clear that $p _3>p _1$.
NOTE
At point $B$, slope of adiabatic (process $B C$ ) is greater than the slope of isothermal (process $A B$ ).





