Heat And Thermodynamics Ques 142
- Starting with the same initial conditions, an ideal gas expands from volume $V _1$ to $V _2$ in three different ways, the work done by the gas is $W _1$ if the process is purely isothermal, $W _2$ if purely isobaric and $W _3$ if purely adiabatic, then
$(2000,2 M)$
(a) $W _2>W _1>W _3$
(b) $W _2>W _3>W _1$
(c) $W _1>W _2>W _3$
(d) $W _1>W _3>W _2$
Show Answer
Solution:
Formula:
- The corresponding $p-V$ graphs (also called indicator diagram) in three different processes will be as shown :
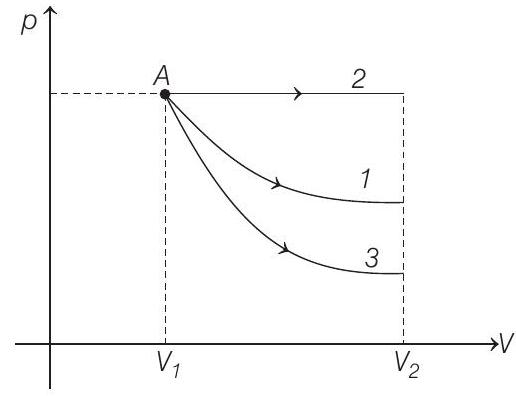
Area under the graph gives the work done by the gas.
$$ \begin{array}{rr} (\text { Area }) _2>(\text { Area }) _1>(\text { Area }) _3 \\ \therefore \quad & W _2>W _1>W _3 \end{array} $$





