Heat And Thermodynamics Ques 21
- A block of ice at $-10^{\circ} C$ is slowly heated and converted to steam at $100^{\circ} C$. Which of the following curves represents the phenomena qualitatively?
$(2000,2 M)$
(a)
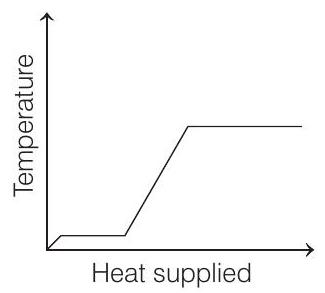
(b)

(c)
(d)

Show Answer
Answer:
Correct Answer: 21.(a)
Solution:
Formula:
- The temperature of ice will first increase from $-10^{\circ} C$ to $0^{\circ} C$. Heat supplied in this process will be
$$ \begin{aligned} & Q _1=m s _i(10) \\ & \text { where, } \quad m=\text { mass of ice } \\ & s _i=\text { specific heat of ice } \end{aligned} $$

Then, ice starts melting. Temperature during melting will remain constant $\left(0^{\circ} C\right)$.
Heat supplied in this process will be
$Q _2=m L, L=$ latent heat of melting.
Now, the temperature of water will increase from $0^{\circ} C$ to $100^{\circ} C$. Heat supplied will be
$$ Q _3=m s _w(100) $$
where, $\quad s _w=$ Specific heat of water.
Finally, water at $100^{\circ} C$ will be converted into steam at $100^{\circ} C$ and during this process temperature again remains constant. Temperature versus heat supplied graph will be as shown in above figure.





