JEE PYQ: Nuclear Physics Question 14
Question 14 - 2020 (05 Sep Shift 1)
Activities of three radioactive substances A, B and C are represented by the curves A, B, and C, in the figure. Then their half-lives $T_1(A) : T_1(B) : T_1(C)$ are in the ratio:
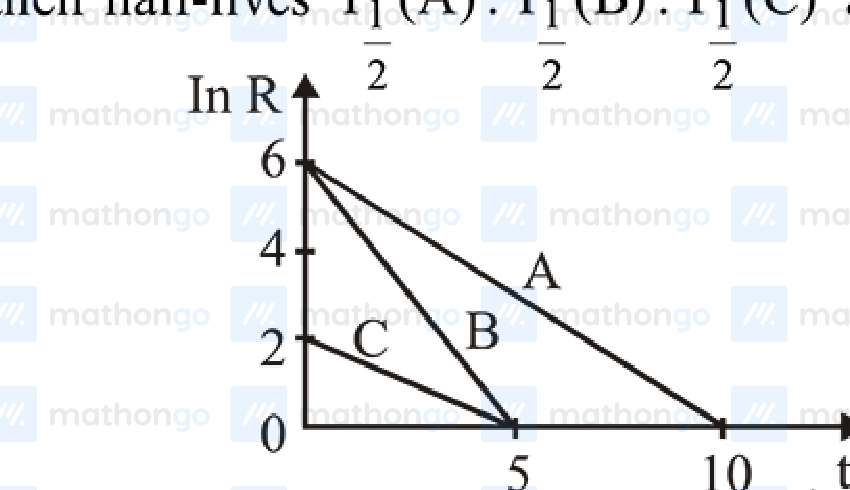
(1) 2 : 1 : 1
(2) 3 : 2 : 1
(3) 2 : 1 : 3
(4) 4 : 3 : 1
Show Answer
Answer: (c) [i.e., (3)]
Solution
Since $R = R_0 e^{-\lambda t}$ $\ln R = \ln R_0 + (-\lambda \ln t)$ $\lambda = \frac{\ln 2}{t_{1/2}} = \text{Slope}$ $\lambda_A = \frac{6}{10} \Rightarrow T_A = \frac{10}{6} \ln 2$ $\lambda_B = \frac{6}{5} \Rightarrow T_B = \frac{5 \ln 2}{6}$ $\lambda_C = \frac{2}{5} \Rightarrow T_C = \frac{5 \ln 2}{2}$ $\therefore T_1 : T_2 : T_3 = \frac{10}{6} : \frac{5}{6} : \frac{15}{6} = 2 : 1 : 3$



