P Block Elements Group 13 To Group 18 Part 3
Topic Importance in JEE
| Metric | Value | Remarks |
|---|---|---|
| Total Questions (2018-2024) | 21 | |
| Weightage | 6.3% | Represents the proportion of questions from this topic. |
Yearly Question Distribution
| Year | Topic Area | Concepts Covered | Number of Questions | Difficulty Level | Key Focus Areas |
|---|---|---|---|---|---|
| 2024 | Group 16 elements | Trends in properties of hydrides / Trends in oxidation states | 2 | Average / Easy | Focus on periodic trends within Group 16. |
| 2023 | Group 13 elements / Group 14 elements / Group 15 elements / Group 17 elements / Group 18 elements | Oxidation state stability of Group 13 elements / Borazine / Uses of Carbon Compounds / Oxoacids of phosphorous / Interhalogen compounds / Uses of helium | 4 | Easy / Average | Broad coverage of multiple p-block groups, emphasizing key compounds and uses. |
| 2022 | Boron family / Group 17 Elements / Group 16 / Group 15 | Structure of diborane / Allotropes of carbon / Stability order of hydrides / Interhalogen compounds / Physical properties of Group 15 | 4 | Average | Structure, allotropy, and stability trends across different p-block groups. |
| 2021 | Group 14 elements: The Carbon Family / Group 18 Elements / Group 17 Elements / Group 16 Elements | Chemical properties / Physical Properties / Oxoacids of halogens / Allotropic forms of Sulphur | 2 | Average / Easy | General properties and allotropy of Group 14, 16, and oxoacids of halogens. |
| 2020 | Group 13 elements / Group 14 elements / Group 15 / Group 16 | Oxide and allotropes of carbon, zeolites / Properties of oxides of carbon / Structure and formulae / Electron gain enthalpy of halogens / Oxoacids of sulphur / Reactions of nitrogen compounds | 3 | Average / Easy | Carbon allotropes and oxides, oxoacids of sulfur, and nitrogen compound reactions. |
| 2019 | Group 14 elements: The Carbon Family / Allotropes of Carbon / Group 13 elements: The Boron Family / Group 15 / Group 16 | Chemical Reactivity / Complex stability / Fullerenes / Inert Pair Effect / Chemical reactivity of Silicon / Thermal stability of hydrides of Group 16 / Preparation processes | 4 | Average / Easy | Reactivity, stability, and inert pair effect across p-block elements, including carbon allotropes. |
| 2018 | Group 13 elements: The Boron Family | Atomic Radii order / Compound Stability / Chemical Reactivity | 2 | Easy | Focus on Group 13 trends in atomic radii, stability, and reactivity. |
Related Video
Study Notes: p-Block Elements – Group 14 (Carbon Family)
Table of Contents
- Introduction to Group 14 Elements
- Key Concepts and Properties
- Important Definitions and Concepts
- Important Compounds and Their Applications
- Comparison of Group 14 Elements
- Examples and Applications
- Summary
1. Introduction to Group 14 Elements
Group 14 elements are also known as the Carbon Family. These elements include:
- Carbon (C)
- Silicon (Si)
- Germanium (Ge)
- Tin (Sn)
- Lead (Pb)
These elements are characterized by having 4 valence electrons and exhibit a range of physical and chemical properties depending on their position in the group.
2. Key Concepts and Properties
2.1 Electronic Configuration
- The general electronic configuration of Group 14 elements is:
[Ne] 3s² 3p² for Carbon
[Ar] 3d¹⁰ 4s² 4p² for Germanium
[Kr] 4d¹⁰ 5s² 5p² for Tin
[Xe] 4f¹⁴ 5d¹⁰ 6s² 6p² for Lead
2.2 Physical Properties
| Element | State at Room Temperature | Melting Point (°C) | Boiling Point (°C) |
|---|---|---|---|
| Carbon | Solid (Graphite) | 3550 | 4827 |
| Silicon | Solid | 1410 | 2960 |
| Germanium | Solid | 938 | 2870 |
| Tin | Solid (Metallic) | 231 | 2275 |
| Lead | Solid (Metallic) | 327 | 1745 |
Note: Carbon in its allotropic form (graphite, diamond) has different physical properties compared to the metallic elements in this group.
3. Important Definitions and Concepts
Definition of Allotropes:
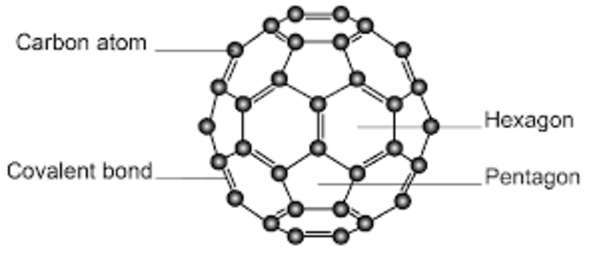
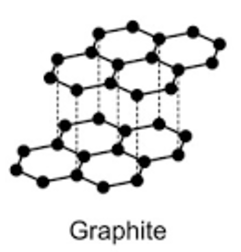
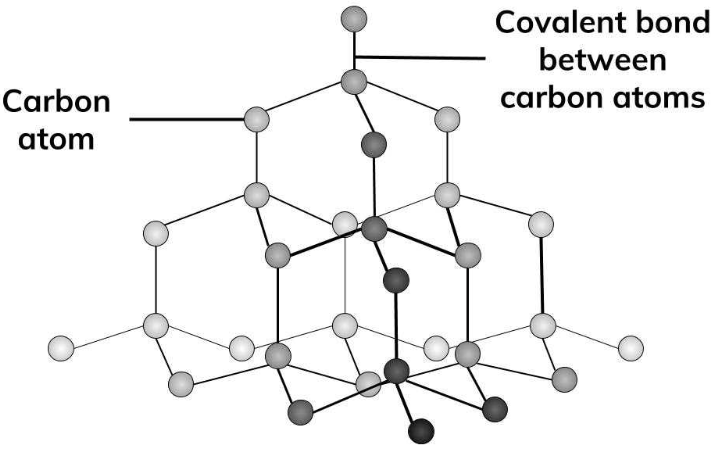
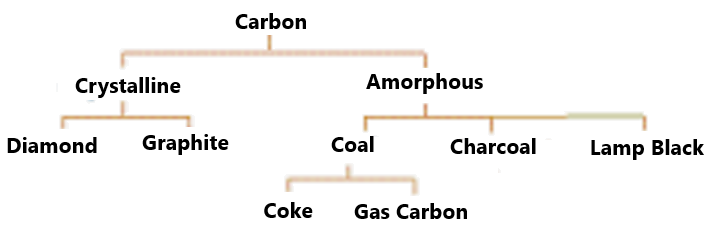
Allotropes are different structural forms of the same element. For example, graphite and diamond are allotropes of carbon.
Definition of Silicate:
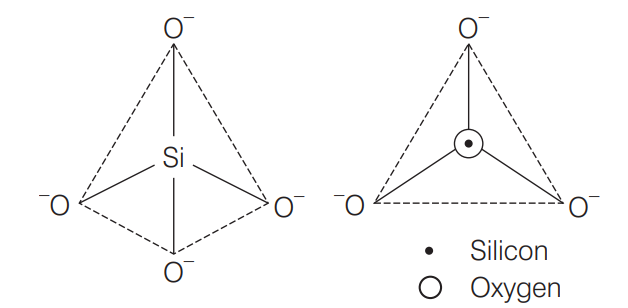
A silicate is a compound containing silicon and oxygen, often in the form of SiO₄⁴⁻ tetrahedral units.
Definition of Zeolites:
Zeolites are porous crystalline aluminosilicates used as catalysts in industrial processes.
4. Important Compounds and Their Applications
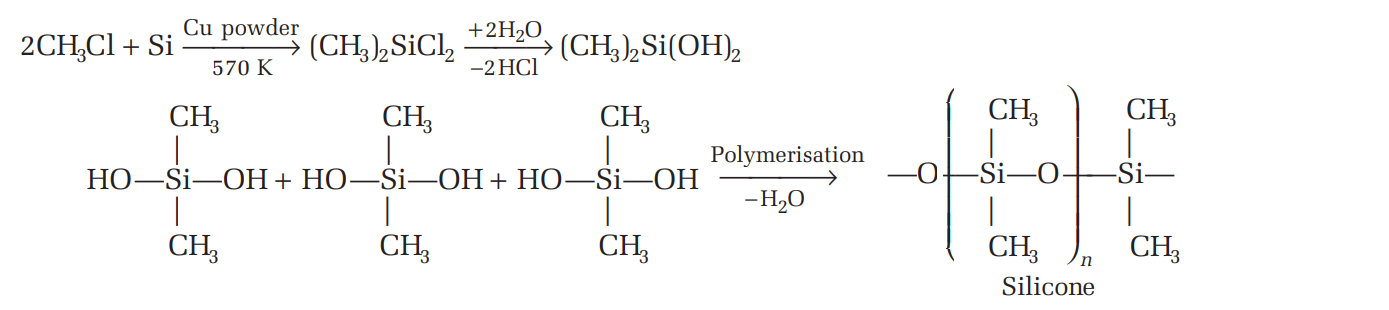
4.1 Silicones
- Definition: Synthetic organosilicon compounds with a repeating unit of R₂SiO.
- Structure: Polymers with alternating Si–O–Si and Si–C bonds.
- Applications: Used in rubber, sealants, adhesives, and medical devices.
4.2 Silicates
- Definition: Compounds containing silicon and oxygen.
- Types:
- Nesosilicates (e.g., Zircon – ZrSiO₄)
- Sorosilicates (e.g., Tourmaline – complex silicate structure)
- Cyclosilicates (e.g., Beryl – Be₃Al₂(SiO₃)₆)
- Inosilicates (e.g., Pyroxene – chains of SiO₄ tetrahedra)
- Phyllosilicates (e.g., Mica – layered structure)
- Tectosilicates (e.g., Quartz – 3D framework of SiO₄ tetrahedra)
Note: Silicates are the most abundant minerals in the Earth’s crust.
4.3 Zeolites
- Definition: Porous, aluminosilicate minerals with a framework structure.
- Properties:
- High surface area
- Ion exchange capacity
- Catalytic activity
- Applications:
- Catalysts in petrochemical industries (e.g., cracking of hydrocarbons)
- Adsorbents in water purification
- Molecular sieves for separating molecules based on size
5. Comparison of Group 14 Elements
| Element | Atomic Number | Atomic Radius (pm) | Electronegativity | Metallic Character | Common Oxidation States |
|---|---|---|---|---|---|
| Carbon | 6 | 77 | 2.55 | Non-metal | +4, +2, +1 |
| Silicon | 14 | 117 | 1.90 | Semi-metal | +4, +2 |
| Germanium | 32 | 122 | 1.81 | Semi-metal | +4, +2 |
| Tin | 50 | 140 | 1.96 | Metal | +4, +2 |
| Lead | 82 | 175 | 1.82 | Metal | +2, +4 |
Note: As we move down the group, metallic character increases, and electronegativity decreases.
6. Examples and Applications
6.1 Carbon
- Graphite: Used in pencils, lubricants, and electrodes.
- Diamond: Used in cutting tools and jewelry.
- Fullerenes: Spherical carbon molecules (e.g., C₆₀) used in nanotechnology.
6.2 Silicon
- Silicon Dioxide (SiO₂): Used in glass, ceramics, and semiconductors.
- Silicon Carbide (SiC): Used in abrasives and high-temperature applications.
6.3 Germanium
- Germanium Dioxide (GeO₂): Used in optical fibers and semiconductors.
6.4 Tin
- Tin Oxide (SnO₂): Used in coatings and pigments.
- Tin Metal: Used in food packaging and soldering.
6.5 Lead
- Lead Oxide (PbO): Used in glass and ceramics.
- Lead Sulfide (PbS): Used in infrared optics.
7. Summary
| Topic | Summary |
|---|---|
| Group 14 Elements | Also known as the Carbon Family, with 4 valence electrons. |
| Physical Properties | Vary from non-metallic (Carbon) to metallic (Lead). |
| Key Compounds | Silicones, Silicates, Zeolites, and their various applications. |
| Applications | Used in electronics, construction, medicine, and industry. |
| Comparisons | Metallic character increases down the group; electronegativity decreases. |
8. Conclusion
Group 14 elements play a crucial role in both natural and synthetic materials. Their unique properties and ability to form a wide range of compounds make them essential in various industries. Understanding the behavior and applications of these elements is vital for students of chemistry and materials science.



