Analytical Chemistry Part 1
Related Video
Study Notes: Qualitative Inorganic Analysis
Table of Contents
- Introduction to Qualitative Inorganic Analysis
- Key Concepts and Definitions
- Common Analytical Techniques
- Important Reactions and Observations
- Summary of Key Tests and Observations
- Conclusion and Final Notes
1. Introduction to Qualitative Inorganic Analysis
Qualitative inorganic analysis is a method used to identify the presence of specific ions or compounds in a sample. This process involves a series of chemical reactions and physical observations to determine the composition of a substance.
2. Key Concepts and Definitions
Qualitative Analysis: The identification of the components of a substance without determining their quantities.
Analyte: The substance being analyzed.
Reagent: A substance added to a sample to cause a chemical reaction that helps identify the analyte.
Precipitation Reaction: A reaction in which two soluble salts react to form an insoluble salt (precipitate).
Color Change: A visual indicator of a chemical reaction or the presence of a specific ion.
Gas Evolution: The release of a gas during a chemical reaction, often used to identify certain anions like carbonate (CO₃²⁻).
3. Common Analytical Techniques
3.1 Precipitation Tests
| Ion | Reagent | Observation | Formula of Precipitate |
|---|---|---|---|
| Cl⁻ | AgNO₃ | White precipitate (AgCl) | AgCl |
| SO₄²⁻ | BaCl₂ | White precipitate (BaSO₄) | BaSO₄ |
| CO₃²⁻ | HCl | Gas evolution (CO₂) | CO₂ |
| OH⁻ | Fe³⁺ | Brown precipitate (Fe(OH)₃) | Fe(OH)₃ |
| S²⁻ | Pb(NO₃)₂ | Black precipitate (PbS) | PbS |
3.2 Flame Tests
| Ion | Flame Color |
|---|---|
| Na⁺ | Yellow |
| K⁺ | Lilac |
| Ca²⁺ | Brick Red |
| Cu²⁺ | Blue Green |
| Li⁺ | Crimson Red |
3.3 Complex Ion Formation
| Ion | Reagent | Observation |
|---|---|---|
| Al³⁺ | NH₃ | White precipitate (Al(OH)₃) |
| Fe³⁺ | NH₃ | Reddish-brown precipitate (Fe(OH)₃) |
| Zn²⁺ | NH₃ | White precipitate (Zn(OH)₂) |
4. Important Reactions and Observations
4.1 Reaction of Carbonate Ions (CO₃²⁻)
- Reagent: Dilute HCl
- Observation: Colorless gas (CO₂) evolves, which turns lime water milky.
- Equation:
$$\text{CO}_3^{2-} + 2\text{H}^+ \rightarrow \text{CO}_2 \uparrow + \text{H}_2\text{O}$$
4.2 Reaction of Chloride Ions (Cl⁻)
- Reagent: AgNO₃ in aqueous solution
- Observation: White precipitate (AgCl) forms.
- Equation:
$$\text{Cl}^- + \text{Ag}^+ \rightarrow \text{AgCl} \downarrow$$
4.3 Reaction of Sulfate Ions (SO₄²⁻)
- Reagent: BaCl₂ in aqueous solution
- Observation: White precipitate (BaSO₄) forms.
- Equation:
$$\text{SO}_4^{2-} + \text{Ba}^{2+} \rightarrow \text{BaSO}_4 \downarrow$$
4.4 Reaction of Sulfide Ions (S²⁻)
- Reagent: Pb(NO₃)₂ in aqueous solution
- Observation: Black precipitate (PbS) forms.
- Equation:
$$\text{S}^{2-} + \text{Pb}^{2+} \rightarrow \text{PbS} \downarrow$$
5. Summary of Key Tests and Observations
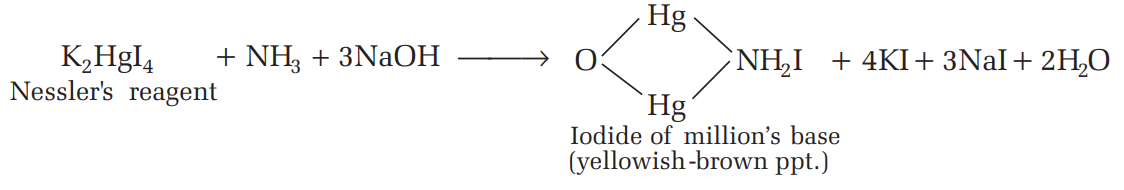
5.1 Common Ions and Their Detection Methods
| Ion | Detection Method | Reagent | Observation |
|---|---|---|---|
| Cl⁻ | Precipitation Test | AgNO₃ | White precipitate |
| SO₄²⁻ | Precipitation Test | BaCl₂ | White precipitate |
| CO₃²⁻ | Gas Evolution Test | HCl | Colorless gas, lime water turns milky |
| OH⁻ | Precipitation Test | Fe³⁺ | Brown precipitate |
| S²⁻ | Precipitation Test | Pb(NO₃)₂ | Black precipitate |
5.2 Flame Test Observations
| Ion | Flame Color |
|---|---|
| Na⁺ | Yellow |
| K⁺ | Lilac |
| Ca²⁺ | Brick Red |
| Cu²⁺ | Blue Green |
| Li⁺ | Crimson Red |
6. Conclusion and Final Notes
Qualitative inorganic analysis relies on a combination of chemical reactions, physical observations, and analytical techniques to identify the components of a sample. By understanding the behavior of different ions under various reagents and conditions, one can effectively determine the composition of an unknown substance.
- Remember: Always conduct experiments in a well-ventilated area and wear appropriate safety gear.
- Practice: Regular practice with different ions and reagents will improve your ability to interpret results.
- Reference: Ensure all reagents are properly labeled and stored to avoid contamination.
Final Notes
- Safety First: Always follow safety protocols when handling chemicals.
- Accuracy Matters: Be precise in recording observations and results.
- Consistency: Maintain consistent procedures for reliable results.
“In qualitative analysis, the art lies not only in knowing what to look for, but also in being able to interpret what you see.”



