Hydrocarbons - Result Question 93
81. One mole of a hydrocarbon $A$ reacts with one mole of bromine giving a dibromo compound, $C _5 H _{10} Br _2$. Compound $A$ on treatment with cold dilute alkaline potassium permanganate solution forms a compound, $C _5 H _{12} O _2$. On ozonolysis $A$ gives equimolar quantities of propanone and ethanal. Deduce the structural formula of $A$.
(1981, 1M)
Show Answer
Solution:
- Ozonolysis products are the key of identification :
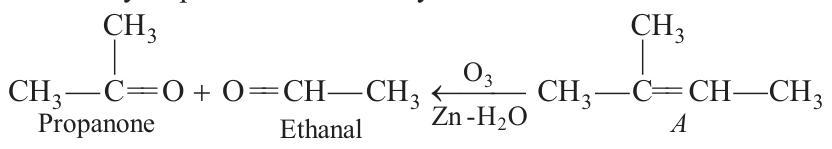
Other products are:





